| ⇦ | 
| ⇨ |
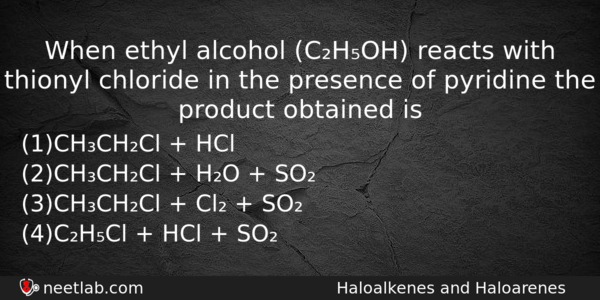
When ethyl alcohol (C₂H₅OH) reacts with thionyl chloride in the presence of pyridine the product obtained is
Options
(a) CH₃CH₂Cl + HCl
(b) CH₃CH₂Cl + H₂O + SO₂
(c) CH₃CH₂Cl + Cl₂ + SO₂
(d) C₂H₅Cl + HCl + SO₂
Correct Answer:
C₂H₅Cl + HCl + SO₂
Explanation:
C₂H₅OH + SOCl₂ → C₂H₅Cl + HCl + SO₂.
Related Questions: - Which of the following will not act as a buffer?
- which of the following non-metal possesses the atomicity double than that
- In which of the following resonance will be possible
- Which of the following statements is correct
- NaOH is prepared by the electrolysis of
Topics: Haloalkenes and Haloarenes
(78)
Subject: Chemistry
(2512)
Important MCQs Based on Medical Entrance Examinations To Improve Your NEET Score
- Which of the following will not act as a buffer?
- which of the following non-metal possesses the atomicity double than that
- In which of the following resonance will be possible
- Which of the following statements is correct
- NaOH is prepared by the electrolysis of
Topics: Haloalkenes and Haloarenes (78)
Subject: Chemistry (2512)
Important MCQs Based on Medical Entrance Examinations To Improve Your NEET Score
18000+ students are using NEETLab to improve their score. What about you?
Solve Previous Year MCQs, Mock Tests, Topicwise Practice Tests, Identify Weak Topics, Formula Flash cards and much more is available in NEETLab Android App to improve your NEET score.
Share this page with your friends

Leave a Reply