| ⇦ | 
| ⇨ |
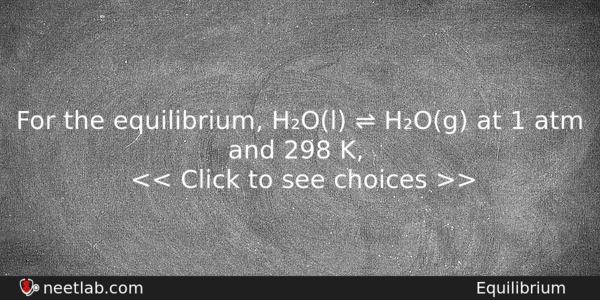
For the equilibrium, H₂O(l) ⇌ H₂O(g) at 1 atm and 298 K,
Options
(a) standard free energy change is equal to zero (ΔG⁰ = 0)
(b) free energy change is less then zero (ΔG⁰ < 0)
(c) standard free energy change is less then zero (ΔG⁰ < 0)
(d) standard free energy change is greater then zero (ΔG⁰ > 0)
Correct Answer:
free energy change is less then zero (ΔG⁰ < 0)
Explanation:
No explanation available. Be the first to write the explanation for this question by commenting below.
Related Questions: - Of the following, which change will shift the reaction towards the product
- A sp³ – hybrid orbital contains
- MnO₃ in an acidic medium dissociates into
- The oxide,which can not act as a reducing agent, is
- In the periodic table, the element with atomic number 16 will be placed
Topics: Equilibrium
(104)
Subject: Chemistry
(2512)
Important MCQs Based on Medical Entrance Examinations To Improve Your NEET Score
- Of the following, which change will shift the reaction towards the product
- A sp³ – hybrid orbital contains
- MnO₃ in an acidic medium dissociates into
- The oxide,which can not act as a reducing agent, is
- In the periodic table, the element with atomic number 16 will be placed
Topics: Equilibrium (104)
Subject: Chemistry (2512)
Important MCQs Based on Medical Entrance Examinations To Improve Your NEET Score
18000+ students are using NEETLab to improve their score. What about you?
Solve Previous Year MCQs, Mock Tests, Topicwise Practice Tests, Identify Weak Topics, Formula Flash cards and much more is available in NEETLab Android App to improve your NEET score.
Share this page with your friends

Leave a Reply