| ⇦ | 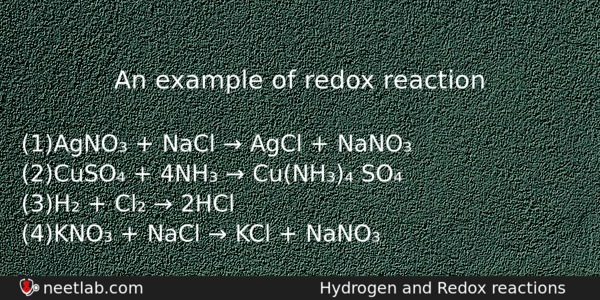
| ⇨ |
An example of redox reaction
Options
(a) AgNO₃ + NaCl → AgCl + NaNO₃
(b) CuSO₄ + 4NH₃ → Cu(NH₃)₄ SO₄
(c) H₂ + Cl₂ → 2HCl
(d) KNO₃ + NaCl → KCl + NaNO₃
Correct Answer:
H₂ + Cl₂ → 2HCl
Explanation:
H₂ + Cl₂ → 2HCl. H₂ (oxidation state = 0 ) changes to H⁺ (oxidation state = +1) and Cl₂ (oxidation state = 0) changes to Cl⁻ (oxidation state = -1). So H₂ has been oxidised and Cl₂ has been reduced, making this is redoxreaction.
Related Questions: - Which one of the following orders is not in accordances with the property
- In which of the following reaction Kp > Kc
- Which of the following is a tribasic acid
- The weight of iron which will be converted into its oxide(Fe₃O₄) by the action of 18g
- Which one of the following compounds will react with NaHCO₃ solution to give
Topics: Hydrogen and Redox Reactions
(174)
Subject: Chemistry
(2512)
Important MCQs Based on Medical Entrance Examinations To Improve Your NEET Score
- Which one of the following orders is not in accordances with the property
- In which of the following reaction Kp > Kc
- Which of the following is a tribasic acid
- The weight of iron which will be converted into its oxide(Fe₃O₄) by the action of 18g
- Which one of the following compounds will react with NaHCO₃ solution to give
Topics: Hydrogen and Redox Reactions (174)
Subject: Chemistry (2512)
Important MCQs Based on Medical Entrance Examinations To Improve Your NEET Score
18000+ students are using NEETLab to improve their score. What about you?
Solve Previous Year MCQs, Mock Tests, Topicwise Practice Tests, Identify Weak Topics, Formula Flash cards and much more is available in NEETLab Android App to improve your NEET score.
Share this page with your friends

Leave a Reply