| ⇦ | 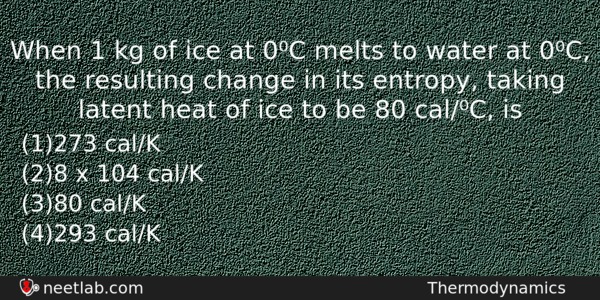
| ⇨ |
When 1 kg of ice at 0⁰C melts to water at 0⁰C, the resulting change in its entropy, taking latent heat of ice to be 80 cal/⁰C, is
Options
(a) 273 cal/K
(b) 8 x 104 cal/K
(c) 80 cal/K
(d) 293 cal/K
Correct Answer:
293 cal/K
Explanation:
Change in entropy is given by
dS = dQ / T or ∆S = ∆Q / T = mLf / 273
∆S = 1000 x 80 / 273 = 293cal / K
Related Questions: - Equal currents are passing through two very long and straight parallel wires
- If a proton, a deuteron and an alpha particle, on being accelerated by the same
- Masses of 3 wires of same metal are in the ratio 1:2:3 and their lengths
- In the half wave rectifier circuit operating from 50 Hz mains frequency,
- A round disc of moment of inertia i₂ about its axis perpendicular to its plane
Topics: Thermodynamics
(179)
Subject: Physics
(2479)
Important MCQs Based on Medical Entrance Examinations To Improve Your NEET Score
- Equal currents are passing through two very long and straight parallel wires
- If a proton, a deuteron and an alpha particle, on being accelerated by the same
- Masses of 3 wires of same metal are in the ratio 1:2:3 and their lengths
- In the half wave rectifier circuit operating from 50 Hz mains frequency,
- A round disc of moment of inertia i₂ about its axis perpendicular to its plane
Topics: Thermodynamics (179)
Subject: Physics (2479)
Important MCQs Based on Medical Entrance Examinations To Improve Your NEET Score
18000+ students are using NEETLab to improve their score. What about you?
Solve Previous Year MCQs, Mock Tests, Topicwise Practice Tests, Identify Weak Topics, Formula Flash cards and much more is available in NEETLab Android App to improve your NEET score.
Share this page with your friends

Leave a Reply