| ⇦ | 
| ⇨ |
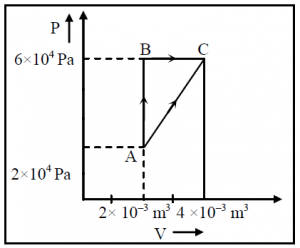
Figure below shows two paths that may be taken by a gas to go from a state A to a state C. In process AB, 400 J of heat is added to the system and in process BC, 100 J of heat is added to the system. The heat absorbed by the system in the process AC will be
Options
(a) 500 J
(b) 460 J
(c) 300 J
(d) 380 J
Correct Answer:
460 J
Explanation:
No explanation available. Be the first to write the explanation for this question by commenting below.
Related Questions: - If A=4i+4j+4k and b=3i+j+4k, then angle between vectors A and B is
- If R is the radius of earth, the height at which the weight of a body 1/4 of its weight
- Two identical capacitors are first connected in series and then in parallel.
- P-type semiconductors are made by adding impurity element
- Length of a conductor is 50 cm radius of cross section 0.1 cm and resistivity
Topics: Thermodynamics
(179)
Subject: Physics
(2479)
Important MCQs Based on Medical Entrance Examinations To Improve Your NEET Score
- If A=4i+4j+4k and b=3i+j+4k, then angle between vectors A and B is
- If R is the radius of earth, the height at which the weight of a body 1/4 of its weight
- Two identical capacitors are first connected in series and then in parallel.
- P-type semiconductors are made by adding impurity element
- Length of a conductor is 50 cm radius of cross section 0.1 cm and resistivity
Topics: Thermodynamics (179)
Subject: Physics (2479)
Important MCQs Based on Medical Entrance Examinations To Improve Your NEET Score
18000+ students are using NEETLab to improve their score. What about you?
Solve Previous Year MCQs, Mock Tests, Topicwise Practice Tests, Identify Weak Topics, Formula Flash cards and much more is available in NEETLab Android App to improve your NEET score.
Share this page with your friends

Leave a Reply