| ⇦ | 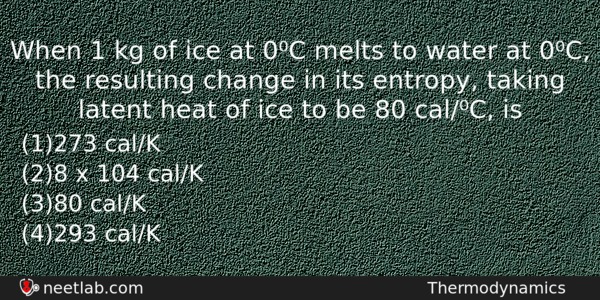
| ⇨ |
When 1 kg of ice at 0⁰C melts to water at 0⁰C, the resulting change in its entropy, taking latent heat of ice to be 80 cal/⁰C, is
Options
(a) 273 cal/K
(b) 8 x 104 cal/K
(c) 80 cal/K
(d) 293 cal/K
Correct Answer:
293 cal/K
Explanation:
Change in entropy is given by
dS = dQ / T or ∆S = ∆Q / T = mLf / 273
∆S = 1000 x 80 / 273 = 293cal / K
Related Questions: - Magnetic flux φ (in weber) linked with a closed circuit of resistance 10 Ω varies
- A total charge of 5µC is distributed uniformly on the surface of the thin walled
- SI unit of permittivity is
- An experiment takes 10 minutes to raise temperature of water from 0⁰C to 100⁰C
- In atom bomb, the reaction which occurs is
Topics: Thermodynamics
(179)
Subject: Physics
(2479)
Important MCQs Based on Medical Entrance Examinations To Improve Your NEET Score
- Magnetic flux φ (in weber) linked with a closed circuit of resistance 10 Ω varies
- A total charge of 5µC is distributed uniformly on the surface of the thin walled
- SI unit of permittivity is
- An experiment takes 10 minutes to raise temperature of water from 0⁰C to 100⁰C
- In atom bomb, the reaction which occurs is
Topics: Thermodynamics (179)
Subject: Physics (2479)
Important MCQs Based on Medical Entrance Examinations To Improve Your NEET Score
18000+ students are using NEETLab to improve their score. What about you?
Solve Previous Year MCQs, Mock Tests, Topicwise Practice Tests, Identify Weak Topics, Formula Flash cards and much more is available in NEETLab Android App to improve your NEET score.
Share this page with your friends

Leave a Reply