| ⇦ | 
| ⇨ |
D(+)-glucose reacts with hydroxyl amine and yields an oxime. The structure of the oxime would be
Options
(a)
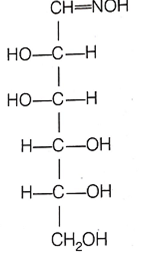
(b)

(c)
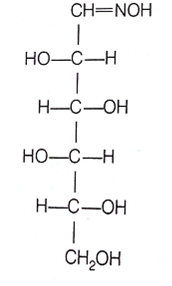
(d)
MISSING
Correct Answer:
Explanation:
No explanation available. Be the first to write the explanation for this question by commenting below.
Related Questions: - The number of isomers in C₄H₁₀O is
- Which one of the following pairs of solution is not an acidic buffer
- The s-block element used as a catalyst in the manufacture of Buna-S rubber is
- Two different gases enclosed in different flasks A and B at same temperature and pressure
- What is the general outer electronic configuration of the coinage metals?
Topics: Biomolecules and Polymers
(88)
Subject: Chemistry
(2512)
Important MCQs Based on Medical Entrance Examinations To Improve Your NEET Score
- The number of isomers in C₄H₁₀O is
- Which one of the following pairs of solution is not an acidic buffer
- The s-block element used as a catalyst in the manufacture of Buna-S rubber is
- Two different gases enclosed in different flasks A and B at same temperature and pressure
- What is the general outer electronic configuration of the coinage metals?
Topics: Biomolecules and Polymers (88)
Subject: Chemistry (2512)
Important MCQs Based on Medical Entrance Examinations To Improve Your NEET Score
18000+ students are using NEETLab to improve their score. What about you?
Solve Previous Year MCQs, Mock Tests, Topicwise Practice Tests, Identify Weak Topics, Formula Flash cards and much more is available in NEETLab Android App to improve your NEET score.
Share this page with your friends

Leave a Reply